Jacksonville Stem Cell Specialist
Stem Cells As A Therapy For Painful Joint, Back, Hand and Foot Conditions.

Advancing The Benefits of Stem Cell Therapy
Dr. Lewis J. Obi is a Jacksonville stem cell specialist and experienced plastic surgeon with decades of pioneering experience in the using a person’s own mesenchymal stem cells to improve painful joint conditions and injuries, spinal conditions and to enhance certain plastic surgery procedures.
Decades of Knowledge and Experience
How Stem Cell Therapy Improves Painful Joint and Back Conditions
ORTHOPEDICS
If you are suffering with chronic knee, shoulder or other joint pain as a result of an injury, osteoarthritis or over use, stem cell therapy may be the answer. Learn more about how stem cells help your body repair itself with your own mesenchymal stem cells without invasive surgery.
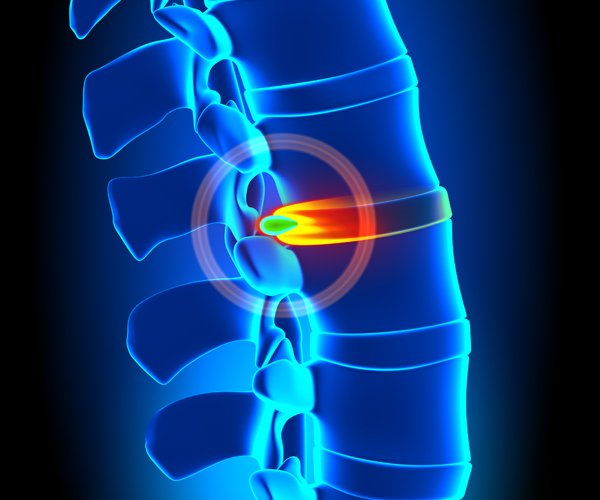
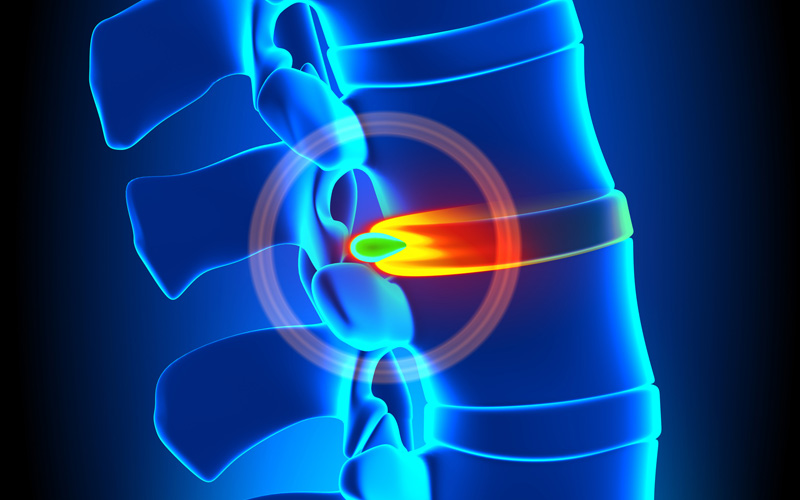
SPINE CONDITIONS
Chronic back pain as a result of an injury or spinal condition can be unbearable. For many, stem cell therapy has lead to significant improvements. If you are living with a painful spinal condition or injury. Help you body repair itself with your own stem cells.
PLASTIC SURGERY
Stem Cells and PRP, (Platelet Rich Plasma) allowed Dr. Obi to provide cosmetic surgery patients with less-invasive procedures, faster recovery times and lasting results. Learn more about how stem cells enhance plastic surgery procedures.
Dr. Obi contributed an entire chapter about the benefits of stem cells in plastic surgery in the first book ever published on stem cells in aesthetic procedures
Dr. Obi contributed an entire chapter to the first book ever published on the use of stem cells to enhance the results of plastic surgery. The chapter offers great detail about how mesenchymal stem cells can be harvested from a patient’s own body fat and used to improve the survival rates of transferred fat, reduce recovery times and many other benefits in the use of adult stem cells in aesthetic procedures.

Why Patients Trusted Dr. Obi
To Be Their Jacksonville Stem Cell Specialist.
In recent years, medical research and development has uncovered the medical benefits of mesenchymal stem cells and demonstrated their value in therapies for a variety of medical conditions. The data from years of research, IRBs and patient results world wide is providing evidence of this.
There are however, many stem cell therapies that are not being performed under IRBs. One example is the use of frozen umbilical cord stem cells. In the U.S., providers of these types of stem cells are required to sterilize them with irradiation. This actually kills the stem cells. Over a period of several years, Dr. Obi regularly examined samples of sterilized umbilical cord stem cells under the microscope, with a NucleoCounter Cell Counter and observed no live stem cells. Also, when we attempted to grow sterilized cord cells, no growth was observed. Other sources of mesenchymal stem cells such bone marrow produce limited quantities of live mesenchymal stem cells which translate into limited results when used for joint and spinal therapy.
Dr. Obi participated in multiple IRBs that documented all of the stem cell therapies he performed and the results for every patient. And these documented results clearly verified that the stem cell therapies he performed improved the joint and back medical conditions in the majority of his patients with no serious complications recorded.
The Correct Quantity and Type of Stem Cells is The Key
When considering stem cell therapy or offering stem cell therapy for patients, it is important to remember that our bodies produce different types of stem cells, and that different stem cells replicate and differentiate to repair and replace damaged and dead cells in different areas of our body. It is also important to understand that there are different ways of harvesting or gathering stem cells, some more effective than others. And quantity matters.
Dr. Obi began harvesting and preparing mesenchymal stem cells using a closed and sterile 38 step process to collect a patient’s own live fat derived mesenchymal adult stem cells. He later refined this process to three steps. This process allowed for the collection and preparation of up to 50 million verified live stem cells from only 2 ounces of body fat.
Mesenchymal stem cells are multipotent stem cells that have the ability to self renew, (replicate) and differentiate into a variety of cell types including bone cells, cartilage cells, muscle cells and fat cells. As a result, they are the correct type of stem cells to serve as a therapy for joint and spine conditions and injuries, and certain plastic surgery procedures that involve fat transfers.
Contact Dr. Obi
Use the contact form to contact Dr. Obi.
Or Call:
(310) 880-3428
Contact Dr. Obi
Use the contact form to contact Dr. Obi.
Or Call!
(310) 880-3428